A recent APSF survey of anesthesia professionals on residual muscle relaxant-induced weakness in the postoperative period was a follow-up of the October 2011 APSF Board of Directors Workshop that addressed future patient safety initiatives (Table 1 on Page 70) by proposing the following 4 questions (https://dev2.apsf.org/newsletters/pdf/winter_2012.pdf).
Responses to Questions Posed at 2011 Workshop
- Do we have evidence/agreement for the etiology of the problem?
- Do we have evidence/agreement for the solution of the problem?
- Does anesthesia have control/influence over introducing the solution to the problem?
- Do we have a way to measure the incidence for baseline and post-intervention data?
At this conference, Sorin J. Brull, MD, addressed “Residual effects of neuromuscular blockers into the postoperative period” as a future APSF safety initiative and proposed the following responses to the 4 questions posed (https://dev2.apsf.org/initiatives.php?id=11)
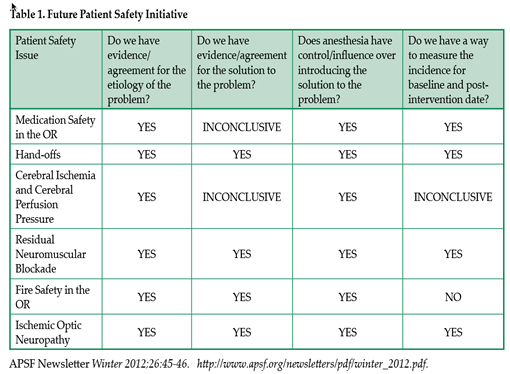
1. Do we have evidence/agreement for the etiology of the problem? YES
We have reports of residual neuromuscular block (residual “curarization”) for over 3 decades (Viby-Mogensen, 1979). The etiology is likely multi-factorial: use of non-depolarizing muscle relaxant agents, lack of intraoperative objective monitoring, and reliance on subjective assessment (visual or tactile means) or clinical tests (head-lift, grip strength, tidal volume, etc) to judge adequacy of pharmacologic reversal prior to tracheal extubation. Recent meta-analysis revealed an incidence of residual paralysis in the PACU of 41% (Naguib, 2007).
2. Do we have evidence/agreement for the solution of the problem? YES
We have many reports that postoperative pulmonary complications continue to occur in patients who experience residual postoperative weakness (Moller, 1990; Pedersen, 1992; Berg, 1997). Patients experience significant delays in meeting PACU and hospital discharge criteria (Murphy, 2004). And we have reports that have documented that appropriate antagonism of neuromuscular block decreases 24-hr morbidity and mortality (Arbous, 2005).
3. Does anesthesia have control/influence over introducing the solution to the problem? YES
In recent U.S. and European surveys, the majority of anesthesiologists (50-65%) believe that postoperative residual weakness is extremely rare (<1%), when in fact the incidence is 41% (Naguib, 2010). For the past decade, studies have shown that perioperative objective neuromuscular monitoring decreases the incidence of postoperative weakness (Mortensen, 1995; Gatke, 2002; Baillard, 2000, 2005; Murphy, 2011).
4. Do we have a way to measure the incidence for baseline and post-intervention data? YES
The currently available neuromuscular monitors (Merck TOF-Watch, GE Healthcare E-NMT, and the Draeger NMT SmartPod) provide objective data on the state of neuromuscular recovery and should be used routinely (Brull, 2010; Murphy, 2011).
With this background, APSF sponsored a panel on monitoring neuromuscular blockade at the 2012 New York Society of Anesthesiologists Postgraduate Assembly (https://dev2.apsf.org/newsletters/pdf/spring2013.pdf) and most recently conducted a survey to determine the opinions of anesthesia professionals with respect to “Residual Muscle Relaxant-Induced Weakness in the Postoperative Period: Is it a Patient Safety Issue?
The announcement of the survey with the link to access the survey was sent to a random sampling of 25% of the active members (including residents and students in training) of the American Society of Anesthesiologists (ASA), American Association of Nurse Anesthetists (AANA) and American Academy of Anesthesiologist Assistants (AAAA) with a follow-up email 14 days after sending the initial announcement. Email announcements were sent to 21,482 anesthesia professionals and 3,182 recipients opened the link to take the survey for a response rate of 14.9% (Figure 1). Nearly 60% of the respondents had been in clinical practice more than 10 years and 72% characterized their clinical practice as the “team model” or a “combination of “solo practitioner and team model.”
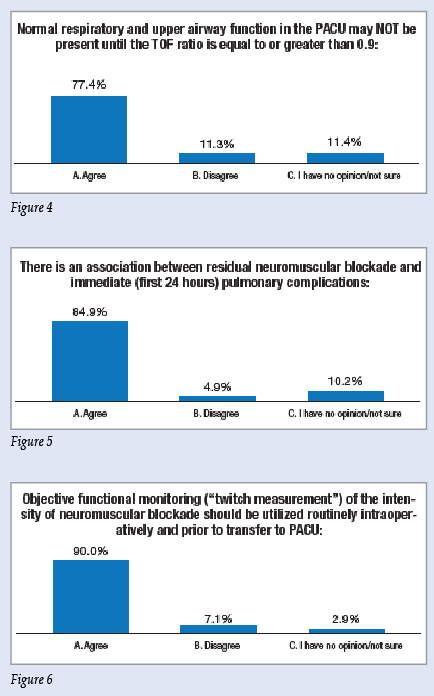
The view as to the frequency of muscle weakness in the PACU due to residual neuromuscular blockade was mixed (Figure 2) but 82% disagreed that a TOF ratio >0.7 confirmed the absence of significant drug-induced neuromuscular weakness in the PACU (Figure 3), and nearly 80% agreed that normal respiratory function may not be present in the PACU until the TOF ratio is equal to or >0.9 (Figure 4). The vast majority of respondents (85%) agreed there was an association between residual neuromuscular blockade and pulmonary complications in the first 24 hours postoperatively (Figure 5) and that appropriate antagonism of neuromuscular blockade would decrease the 24-hour major postoperative morbidity and mortality.
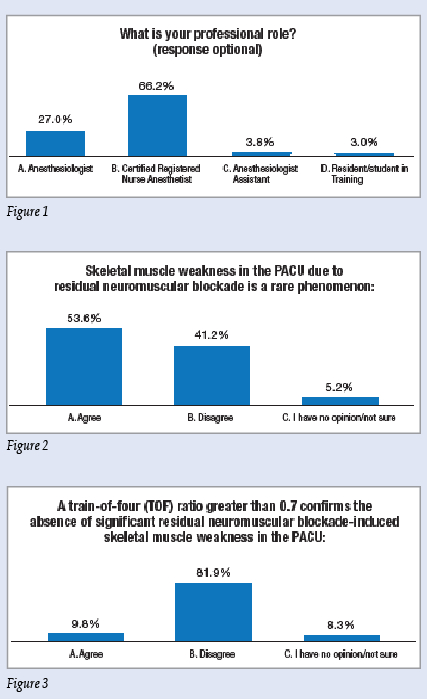
 Figures 1-6: Responses from 3,182 out of 21,482 randomly sampled anesthesia professionals from the APSF Survey “Residual Muscle Relaxant-Induced Weakness in the Postoperative Period: Is it a Patient Safety Issue?
Figures 1-6: Responses from 3,182 out of 21,482 randomly sampled anesthesia professionals from the APSF Survey “Residual Muscle Relaxant-Induced Weakness in the Postoperative Period: Is it a Patient Safety Issue?
Ninety per cent of the respondents agreed that objective functional monitoring (twitch measurement) should be utilized routinely intraoperatively for patients receiving nondepolarizing neuromuscular blocking drugs prior to transfer to the PACU (Figure 6). Consistent with this view nearly 80% of the survey responses supported the statement that “APSF should encourage professional associations (ASA, AANA, AAAA) to consider adding “Monitoring Neuromuscular Function” to their “standards/recommendations” for the intraoperative care of those patients receiving neuromuscular blocking drugs.
Robert K. Stoelting, MD President, APSF
APSF thanks Lorraine Jordan, CRNA, PhD, AANA Senior Director of Research and AANA Foundation Executive Director, for creation of the graphics depicting the survey responses.


 Issue PDF
Issue PDF